23 . 11 . 2018
Fecal Microbiota Transplantation - Potential Therapeutic Uses
In recent years, the scientific community has recognized the intestinal microbiome’s important role in various metabolic and immunological processes. The word microbiome refers to the ecosystem of microorganisms present in the human body, including bacteria, viruses and fungi. Each human being has its unique and characteristic ecosystem that contributes to a multitude of physiological reactions.
There are several ways to change the intestinal microbiome, such as: elimination diets, probiotics, prebiotics, antibiotics and fecal microbiota transplantation (FMT). The latter is the most radical approach since it consists of directly inoculating a donor’s microbiota into the recipient’s gut. Interestingly, the first records of FMT have been traced back to the fourth century, in China, where human fecal material was used in patients with severe diarrhea, although the technique used was quite rudimentary.
What is the main indication for fecal transplantation?
Currently, the major indication for FMT is recurrent pseudomembrane colitis, a severe intestinal infection caused by the bacteria Clostridium difficile. In the United States, FMT was approved for this indication in 2013 and has a cure rate of about 90%, which is much higher than the 20-30% achieved with antibiotic therapy.
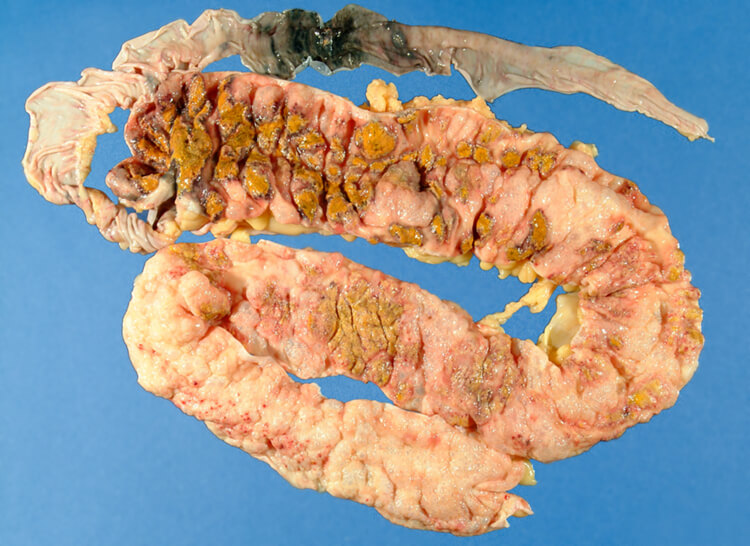
Fig. 1 – Pseudomembranous colitis is a life threatening infection of the intestine caused by Clostridium difficile, and it may be refractory to antibiotics.
What other potential indications can FMT have?
Given the potential of directly altering the intestinal flora of an individual, the majority of studies have been confined to the gastrointestinal tract, specifically for the following conditions:
– Functional gastrointestinal disorders – conditions where there are no structural anatomical changes, despite the presence of symptoms. Irritable bowel syndrome and chronic constipation belong to this category.
– Inflammatory bowel disease – severe pathology due to an aberrant immune response to antigens expressed by the intestinal microbiome. Crohn’s disease and ulcerative colitis are the two types of inflammatory bowel disease.
However, given the known implications of the microbiome in systemic processes, other potential areas of research are being explored, such as the metabolic syndrome and type 2 diabetes mellitus, obesity, autoimmune diseases, neurodegenerative diseases, among others.
What do the currently available studies tell us about its effectiveness?
In contrast to the great effectiveness in treating recurrent Clostridium difficile colitis, the remission rates of inflammatory bowel disease, in the studies published so far, are much less impressive. A 2014 meta-analysis showed a remission rate of 36.2% (greater in Crohn’s disease than in ulcerative colitis). Another randomized controlled study of 2015 on 50 patients with ulcerative colitis did not show any difference in post-transplantation clinical or endoscopic remission rates.
In irritable bowel syndrome, results have also been mixed, not documenting a clear therapeutic efficacy. In turn, a Chinese study in 276 patients with chronic constipation showed improvement in 67% and complete cure in 40% of the cases.
How is FMT performed?
First and foremost, the donor must be carefully selected. Several questionnaires about lifestyle, eating habits, medical history and previous antibiotic use are gathered. In order to rule out the presence of infectious agents, blood and stool tests are performed. The fecal content is then processed and delivered via the upper GI route (nasogastric tube or capsules) or lower GI route (colonoscopy or retention enemas).
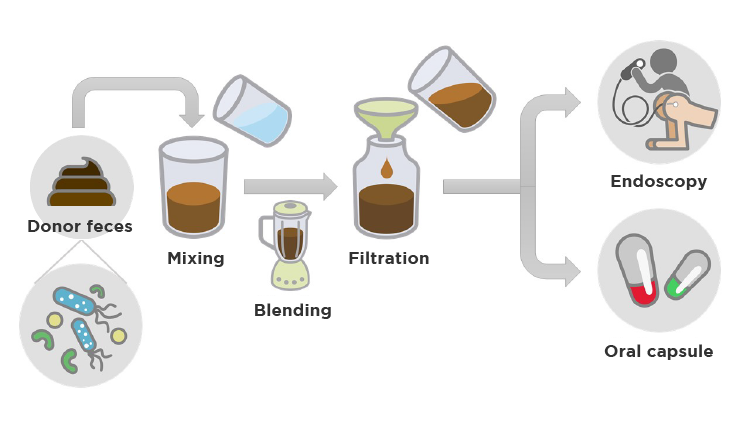
Fig.2 – Schematic diagram of the fecal microbiota transplantation process.
Is it a safe procedure?
Side effects are usually gastrointestinal and transient (changes in bowel movements, bloating, abdominal pain, etc.). However, when the procedure involves endoscopic procedures, there is always the risk associated with these techniques. Since FMT use is relatively recent, it is not possible to know potential long-term side effects yet. It is important to note that, although there is already a solid body of knowledge about the mlcrobiome’s batterial component, viruses are more difficult to identify and characterize, leaving some room for potential flaws in the current screening methods.
FMT has a fascinating therapeutic potential and puts to the tests all the theories that have been developed around the microbiome. However, the FMT clinical trials do not prove it to be a therapeutic “miracle” so far, with exception for the recurrent Clostridium difficile infection. Maybe as our knowledge about the gut microbiome expands, it will be possible to identify which microorganisms are good for each particular condition and then select donors accordingly. This would make the transplantation process directed and more effective. Undoubtedly, we will have many exciting developments in this area of research in the near future.
References:
Wang JW, Kuo CH, Kuo FC, et al. Fecal microbiota transplantation: Review and update. Journal of the Formosan Medical Association. 2018.
Sunkara T, Rawla P, Ofosu A, Gaduputi V. Fecal microbiota transplant – a new frontier in inflammatory bowel disease. Journal of Inflammation Research. 2018;11:321-328.
Schmulson M, Bashashati M. Fecal microbiota transfer for bowel disorders: efficacy or hype? Current Opinion in Pharmacology. 2018;43:72-80.
Colman RJ, Rubin DT. Fecal microbiota transplantation as therapy for inflammatory bowel disease: a systematic review and meta-analysis. Journal of Crohns and Colitis. 2014;8(12):1569–1581.
Rossen NG, Fuentes S, van der Spek MJ, et al. Findings from a randomized controlled trial of fecal transplantation for patients with ulcerative colitis. Gastroenterology. 2015;149(1):110.e4–118.e4.
Li N, Tian H, Ma C et al. Efficacy analysis of fecal microbiota transplantation in the treatment of 406 cases with gastrointestinal disorders. Chinese Journal of Gastrointestinal Surgery 2017, 20:40-46.